Is Matter Around Us Pure - NCERT Questions
What is meant by a pure substance?
SOLUTION:A pure substance is one which is made up of only one kind of atoms or molecules. e.g., water is made up of only one kind of particles. So water is a pure substance.
Q 2.List the points of difference between homogeneous and heterogeneous mixture.
SOLUTION: 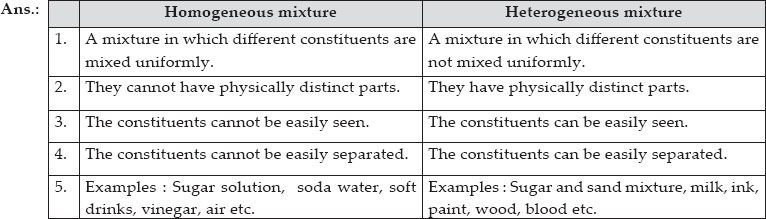
Differentiate between homogeneous and heterogeneous mixtures with examples.
SOLUTION:Refer Ans. 2 above.
Q 4.How are sol, solution and suspension different from each other?
SOLUTION: 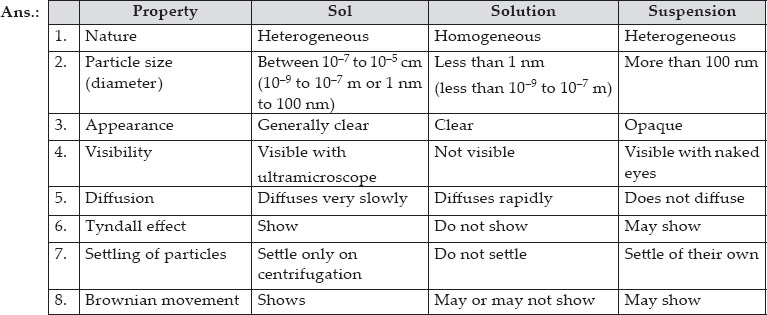
To make a saturated solution 36 g of sodium chloride is dissolved in 100 g of water at 293 K. Find its concentration at this temperature.
SOLUTION:Mass of sodium chloride = 36 g
Mass of solution = 36 + 100 = 136 g
Concentration of solution
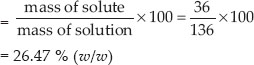
= 26.47 % (w/w)
How will you separate a mixture containing kerosene and petrol. (difference in their boiling points is more than 25°C), which are miscible with each other?
SOLUTION: The mixture of two miscible liquids such as kerosene and petrol whose boiling points differ by more than 25°C can be easily separated by the technique of simple distillation.
At the boiling point of more volatile (low boiling) liquid of the mixture, the vapours almost exclusively consist of the more volatile liquid. Likewise at the boiling point of the less volatile (high boiling) liquid, vapours almost entirely consist of the less volatile liquid since the more volatile liquid has already distilled over.
Name the technique to separate
(I) butter from curd
(ii) salt from sea water
(iii) camphor from salt.
(I) Butter from curd can be separated by the technique of centrifugation.
(ii) Salt from sea water can be separated by the technique of crystallisation or by the evaporation.
(iii) Camphor is sublimable but salt is not. So, camphor can be separated from salt by sublimation technique.
What type of mixtures are separated by the technique of crystallisation?
SOLUTION:Homogeneous mixtures such as common salt solution and copper sulphate solution are separated by technique of crystallisation.
Q 9. Classify the following as chemical or physical changes.
(1) cutting of trees
(2) melting of butter in a pan
(3) rusting of almirah
(4) boiling of water to form steam
(5) passing of electric current through water and the water breaking down into hydrogen and oxygen
(6) dissolving common salt in water
(7) making a fruit salad with raw fruits
(8) burning of paper and wood
(1) Cutting of trees is a chemical change since all chemical reactions stop and we cannot get back the original tree from the wooden pieces.
(2) Melting of butter in a pan is a physical change since there is no change in the chemical composition of butter, only the physical state changes from solid to liquid.
(3) Rusting of almirah is a chemical change since during rusting, a new chemical compound called hydrated iron oxide (rust) is formed.
(4) Boiling of water to form steam is a physical change because during this change only change of state occurs from liquid water to steam (gaseous) without any change in its chemical composition.
(5) Passing of electric energy through water to form hydrogen and oxygen is a chemical change since the properties of hydrogen (combustible gas) and oxygen (supporter of combustion) are altogether different from those of water which is neither combustible nor a supporter of combustion but it actually extinguishes fire.
(6) Dissolution of common salt in water is a physical change since salt can be easily recovered by evaporating water.
(7) Making a fruit salad with raw fruits is a physical change since there is no change in the chemical properties of the fruits but only the physical appearance has changed.
(8) Burning of paper is a chemical change since carbon dioxide, water vapours, smoke and ash which are the products of combustion cannot be converted back into paper or wood by any physical method.
Try segregating the things around you as pure substances or mixtures.
SOLUTION:Pure substances : Distilled water, diamond, graphite, gold, sulphur
Mixtures : Curd, ice cream, kerosene oil, cooking oil, steel, vulcanised rubber, solder wire (alloy of lead and tin).
Which separation techniques will you apply for the separation of the following?
(A) Sodium chloride from its solution in water.
(B) Ammonium chloride from a mixture containing sodium chloride and ammonium chloride.
(C) Small pieces of metal in the engine oil of a car.
(D) Different pigments from an extract of flower petals.
(E) Butter from curd.
(F) Oil from water.
(G) Tea leaves from tea.
(H) Iron pins from sand.
(I) Wheat grains from husk.
(J) Find mud particles suspended in water.
(A) Evaporation : Water will evaporate leaving behind sodium chloride.
(B) Sublimation : Ammonium chloride will be collected as sublimate.
(C) Filtration : Pieces of metal can be separated by filtration.
(D) Chromatography : Pigments (coloured components) from the extract of flower plants can be separated by chromatography.
(E) Centrifugation : Butter will get separated upon centrifugation.
(F) Separating funnel : Oil and water can be separated by the use of separating funnel.
(G) Filtration : Upon filtration through a sieve, tea leaves will be collected on the sieve.
(H) Magnetic separation : A magnet will attract iron pins and not sand particles.
(I) Sieving : Wheat grains from husk can be separated with the help of sieves.
(J) Sedimentation : As a result of sedimentation, mud particles will settle as precipitate. It can be separated later on by filtration.
Write the steps you would use for making tea. Use the words solution, solvent, solute, dissolve, soluble, insoluble, filtrate and residue.
SOLUTION:Take 100 mL of water which acts as solvent. Boil water on a gas stove. Add one teaspoon of sugar which acts as solute. Sugar is soluble in water, so gets dissolved in water and forms a solution. Now add about half a teaspoon of tea leaves which are insoluble in water. Boil the contents for 4 to 5 minutes and add a half cup of milk and allow to boil again for 2-3 minutes. Filter the tea with the help of a sieve. Tea leaves will be left as residue while tea will be obtained as filtrate.
Q 13.Pragya tested the solubility of three different substances at different temperatures and collected the data as given below (results are given in the following table, as grams of substance dissolved in 100 grams of water to form a saturated solution).
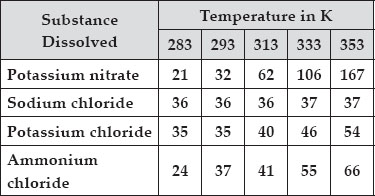
(A) What mass of potassium nitrate would be needed to produce a saturated solution of potassium nitrate in 50 grams of water at 313 K?
(B) Pragya makes a saturated solution of potassium chloride in water at 353 K and leaves the solution to cool at room temperature. What would she observe as the solution cools? Explain.
(C) Find the solubility of each salt at 293 K. Which salt has the highest solubility at this temperature?
(D) What is the effect of change of temperature on the solubility of a salt?
(A) Solubility of potassium nitrate at 313 K
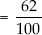
∵ 100 g of water contains potassium nitrate
= 62 g
∴ 50 g of water contains potassium nitrate
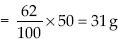
Thus, 31 g potassium nitrate would be needed to produce a saturated solution of potassium nitrate in 50 g of water at 313 K.
(B) When a saturated solution of potassium chloride at 353 K is cooled, the solubility of potassium chloride in water decreases. As a result the amount of potassium chloride which exceeds its solubility at lower temperature separates out as crystals.
(C) The maximum amount of the salt which is dissolved in 100 g of water to form a saturated solution at the given temperature, is known as solubility.

From the above data, it is clear that ammonium chloride has the highest solubility at 293 K.
(D) In general, the solubility of a salt increases with temperature. The increase is, however, different for different salts. For example, the solubility of potassium nitrate increases appreciably, that of ammonium chloride increases slightly, that of potassium chloride increases marginally while that of sodium chloride almost remains constant.
Explain the following giving examples.
(A) saturated solution
(B) pure substance
(C) colloid
(D) suspension
(A) Saturated solution : A solution in which no more solute can be dissolved in a given amount of solvent at a particular temperature is called saturated solution. For example, if we dissolve 40 g sodium chloride in 100 g of water at 293 K, it will form a saturated solution because the solubility of sodium chloride at 293 K is 36 g per 100 g of water.
(B) Pure substance : A substance made up of only one kind of atoms or molecules is called a pure substance. A pure substance has the same colour, taste and texture at a given temperature and pressure. A pure substance also has a fixed melting and boiling point at a constant pressure. For example, hydrogen gas, sodium chloride, water, etc.
(C) Colloid : A substance is said to be a colloid if the particle size lies between 1 to 100 nm. A colloidal solution is heterogeneous and consists of two phases. i.e., dispersed phase (colloidal particles) and dispersion medium in which colloidal particles are suspended. For example, colloidal solution of sulphur or starch, milk, etc.
(D) Suspension : It is a heterogeneous mixture in which the particles of the solute do not dissolve but remain suspended throughout the bulk of the solvent. The size of the suspension particles is more than 10–7 m. For example, chalk powder in water is a suspension.
Classify each of the following as a homogeneous or heterogeneous mixtures.Soda water, wood, air, soil, vinegar, filtered tea
SOLUTION:Homogeneous mixtures : Soda water, vinegar and filtered tea.
Heterogeneous mixtures : Wood and soil.
Air is a homogeneous mixture of different gases. However, if some dust or other particles are also present, then air becomes heterogeneous mixture.
How would you confirm that a colourless liquid given to you is pure water?
SOLUTION:If the boiling point and freezing point of the given liquid comes out to be 100°C or 373 K and 0°C or 273 K respectively under one atmospheric pressure, it confirms that the given liquid is pure water.
Q 17.Which of the following materials fall in the category of a “pure substance”
SOLUTION:Ice, iron, calcium oxide and mercury are pure substances since they contain particles of only one kind of matter. In contrast, milk, hydrochloric acid (hydrogen chloride gas dissolved in water), brick and air cannot be called pure substances because they consists of particles of more than one kind of matter.
Q 18. Identify the solutions among the following mixtures.
(A) Soil,
(B) sea water,
(C) air,
(D) coal,
(E) soda water
A solution is a homogeneous mixture of two or more substances. In the light of this, the solutions among the given mixtures are (B) sea water, (C) air and (E) soda water.
Q 19. Which of the following will show Tyndall effect?
(A) Salt solution
(B) Milk
(C) Copper sulphate solution
(D) Starch solution
(B) milk and (D) starch solution show Tyndall effect because they are colloidal solutions, whereas (A) salt solution and (C) copper sulphate solution are true solutions. Their particle size is too small to scatter light, so they do not show Tyndall effect.
Q 20.Classify the following into elements, compounds and mixtures.
(A) Sodium,
(B) soil,
(C) sugar solution,
(D) silver,
(E) calcium carbonate,
(F) tin,
(G) silicon,
(H) coal,
(I) air,
(J) soap,
(K) methane,
(L) carbon dioxide,
(M) blood
Elements : The elements are regarded as the building blocks of the universe. So (A) sodium, (D) silver, (F) tin and (G) silicon are elements.
Compounds : It is a pure substance made up of two or more elements chemically combined in a fixed proportion by mass. So, (E) calcium carbonate, (K) methane and (L) carbon dioxide are compounds.
Mixtures : It is a substance containing two or more substances (elements or compound) in any proportion. So (B) soil, (C) sugar solution, (H) coal, (I) air, (J) soap and (M) blood are mixtures.
Which of the following are chemical changes?
(A) Growth of a plant
(B) Rusting of iron
(C) Mixing of iron filings and sand
(D) Cooking of food
(E) Digestion of food
(F) Freezing of water
(G) Burning of a candle
(A) Growth of a plant
(B) rusting of iron,
(D) cooking of food
(E) digestion of food and
(G) burning of a candle, are chemical changes.