Periodic Classification of Elements - NCERT Questions
Did Doberiener's triads also exist in the columns of Newland's octaves? Compare and find out.
SOLUTION:Ans.: Yes, some of the Doberiener's triads did exist in the columns of Newlands' octaves. For example,
Q 2.What were the limitations of Dobereiner's classification ?
SOLUTION:Ans.: Dobereiner could classify only three triads from the elements known at that time. It is not found useful because all the elements known at that time could not be arranged in triads.
Q 3.What were the limitations of Newlands' Law of Octaves?
SOLUTION:Ans.: The major limitations of Newlands' law of octaves were :
(i) It was applicable to only lighter elements having atomic masses upto 40 u, i.e., upto calcium. After calcium, the first and the eighth element did not have similar properties. For example chromium (Cr) and yttrium (Y) are the first and the eighth element placed in the same column but they have entirely different properties.
(ii) It was assumed that only 63 elements existed in nature and no new elements would be discovered in the future. But later on several new elements were discovered whose properties did not fit into the law of octaves.
(iii) Some similar elements have been separated from one another while some dissimilar elements have been placed in the same column. For example, iron (Fe) resembles cobalt (Co) and nickel (Ni) but has been placed far away from these elements. Similarly, cobalt and nickel do not resemble halogens (F, Cl, Br) but have been placed in the same column.
(iv) When noble gases were discovered, the properties of the eighth element were no longer similar to the first one. Actually, now it was the first and the ninth element which has similar properties.
Use Mendeleev's periodic table to predict the formulae for the oxides of the following elements: K, C, Al, Si, Ba.
SOLUTION:Ans.: Oxygen is a member of group VI A in Mendeleev's periodic table. Its valency is 2. Similarly, the valencies of all the elements listed can be predicted from their respective groups. This can help in writing the formulae of their oxides.
(i) Potassium (K) is a member of group IA. Its valency is 1. Therefore, the formula of its oxide is K2O.
(ii) Carbon (C) is a member of group IV A. Its valency is 4. Therefore, the formula of its oxide is C2O4 or CO2.
(iii) Aluminium (Al) belongs to groups III A and its valency is 3. The formula of its oxide is Al2O3.
(iv) Silicon (Si) is present in group IV A after carbon. Its valency is also 4. The formula of its oxide is Si2O4 or SiO2.
(v) Barium (Ba) belongs to group II A and the valency of the element is 2. The formula of the oxide of the element is Ba2O2 or BaO.
Besides gallium, which other elements have since been discovered that were left by Mendeleev in his periodic table? (any two)
SOLUTION:Ans.: Germanium, Scandium.
Q 6.What were the criteria used by Mendeleev in creating his periodic table?
SOLUTION:Ans.: Mendeleev used atomic masses of the elements as the criteria for creating his periodic table. In this table, the elements were arranged in order of increasing atomic masses.
Q 7.Why do you think the noble gases are placed in a separate group?
SOLUTION:Ans.: Out of all the elements known, noble gases such as helium (He), neon (Ne), argon (Ar), krypton (Kr) and xenon (Xe) are the most inert (unreactive) and are present in extremely low concentrations in our atmosphere. Therefore, they are grouped together in a separate group called zero group in the Mendeleev's periodic table.
Q 8.How could the modern periodic table remove various anomalies of Mendeleev periodic table?
SOLUTION:Ans.: The modern periodic table removes all the anomalies and drawbacks of the Mendeleev's periodic table based on the increasing atomic mass by changing the basis of classification to atomic number. For example,
(a) All the isotopes of an element are now placed in the same position because they all have the same atomic number.
(b) The anomaly of higher atomic mass element being placed before the lower atomic mass element is removed when these elements are arranged in the order of increasing atomic number.
Name two elements you would expect to show chemical reactions similar to magnesium. What is the basis for your choice?
SOLUTION:Ans.: Magnesium (Mg) belongs to group 2 known as alkaline earth family. The two other elements belonging to the same group are calcium (Ca) and strontium (Sr). The basis of choice is the electronic distribution in the valence shell of these elements. All of them have two electrons each. For example :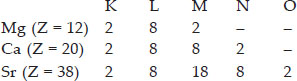
Name :
A three elements that have a single electron in their outermost shells.
B. two elements that have two electrons in their outermost shells.
C. three elements with filled outermost shells.
Ans. (A) (i) Lithium (ii) Sodium (iii) Potassium
(B) (i) Beryllium (ii) Magnesium
(C) (i) Helium (ii) Neon (iii) Argon.
A. Lithium, sodium, potassium are all metals that react with water to liberate hydrogen gas. Is there any similarity in the atoms of these elements?
B. Helium is an unreactive gas and neon is a gas of extremely low reactivity. What, if anything, do their atoms have in common?
Ans.: (A) Lithium, sodium and potassium all react with water to form alkalies, i.e., lithium hydroxide, sodium hydroxide, potassium hydroxide, etc. with the liberation of hydrogen gas.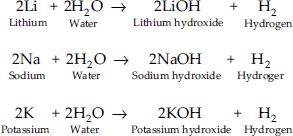
All these metals have one electron in their respective outermost shells.
(B) Helium and neon are noble gases and hence have extremely low chemical reactivity. The common thing in these gases is that they have their shells completely filled. Helium has only K-shell which is complete, i.e., has 2 electrons. Neon, on the other hand, have two shells, K and L. Both these shells are complete, i.e., K shell has 2 electrons and L shell has 8 electrons.
In the modern periodic table, which are the metals among the first ten elements?
SOLUTION:Ans.: Metals among the first ten elements are lithium (Li) and beryllium (Be). These are placed towards the left of the table.
Q 13. By considering their position in the periodic table, which one of the following elements would you expect to have maximum metallic characteristics?
Ga, Ge, As, Se, Be
Ans.: Arranging the above elements in different groups and periods in order of their increasing atomic numbers, we have
Periods 1 2 3 4
Group 1 – – – –
Group 2 – Be – –
Group 13 – – – Ga
Group 14 – – – Ge
Group 15 – – – As
Group 16 – – – Se
We know that metallic character decreases from left to right in a period and increases down a group. Therefore, out of the elements listed in the question, Be and Ga are expected to be most metallic. Out of Be and Ga, Ga is bigger in size and hence has greater tendency to lose electrons than Be. Therefore, Ga is more metallic than Be.
Which of the following statements is not a correct statement about the trends when going from left to right across the periods of periodic table?
A. The elements become less metallic in nature.
B. The number of valence electrons increases.
C. The atoms lose their electrons more easily.
D. The oxides become more acidic.
Ans.: (C) The atoms lose their valence electrons with difficulty and not easily. This is on account of the reasons that
• nuclear charge increases from left to the right since the atomic number of the elements gradually increases.
• with the increase in nuclear charge, the force binding the electrons with the nucleus increases.
Therefore, the atoms lose their valence electrons with difficulty.
Element X forms a chloride with the formula XCl2 which is a solid with high melting point. X would most likely to be in the same group of the periodic table as :
A. Na
B. Mg
C. Al
D. Si
Ans.: (B) The formula of the chloride of the element is XCl2. This means that the valency of the element X is 2 since chlorine is monovalent. The element with valency 2 is expected to be present in group 2 to which magnesium (Mg) belongs.
Q 16. Which element has
A. two shells, both of which are completely filled with electrons ?
B. the electronic configuration 2, 8, 2?
C. a total of three shells, with four electrons in its valence shell?
D. a total of two shells, with three electrons in its valence shell?
E. twice as many electrons in its second shell as in its first shell ?
Ans.: (A) Neon, Ne (2, 8)
(B) Magnesium, Mg (2, 8, 2)
(C) Silicon, Si (2, 8, 4)
(D) Boron, B (2, 3)
(E) Carbon, C (2, 4)
A. What property do all electrons in the same column of the periodic table as boron have in common ?
B. What property do all elements in the same column of the periodic table as fluorine have in common ?
Ans.: (A) All the elements which lie in the same column as that of boron belong to group 13. Therefore, they have three electrons in their respective valence shells. Except, boron which is a non-metal, all other elements (i.e., aluminium, gallium, indium and thallium) in this group are metals.
(B) The elements which lie in the same column as fluorine are called halogens. They belong to group 17 and thus have seven electrons in the valence shell. Therefore, their valency is 18 – 17 = 1. All these elements (fluorine, chlorine, bromine, iodine and astatine) are non-metals.
An atom has electronic configuration 2, 8, 7.
A. What is the atomic number of this element?
B. To which of the following elements would it be chemically similar ? (Atomic numbers are giving in parentheses).
N(7), F (9), P(15), Ar(18)
Ans.: (A) The atomic number of the element is 17 (2 + 8 + 7 = 17).
(B) It will be chemically similar with fluorine (F) which has also 7 electrons in valence shell (2, 7).
The position of three elements A, B and C in the periodic table are shown below :
| Group 16 | Group 17 |
| – | – |
| – | A |
| – | – |
| B | C |
A. State whether A is metal or non-metal.
B. State whether C is more reactive or less reactive than A.
C. Will C be larger or smaller in size than B?
D. Which type of ion, cation or anion will be formed by the element A? SOLUTION:
Ans.: (A) Group 17 represents halogen family. All the elements included in the family are non-metals. Therefore, element A is a non-metal.
(B) Reactivity of non-metals is generally due to the electron accepting tendency of their atoms. Down the group, the atomic size increases. Therefore, the attraction of the nucleus for the outside electrons decreases. This means that down the group of non-metals, reactivity decreases. Thus, the element C is less reactive than the element A.
(C) Atomic size of the elements decreases along a period. The elements B and C are present in the same period. Since C is placed after B, the size of the element C is less than that of B.
(D) The element A, as pointed out earlier is a non-metal which belongs to group 17. It has seven valence electrons (2, 8, 7). In order to have the configuration of the nearest noble gas element, it will take up one electron and change to anion i.e., A– ion.
Nitrogen (atomic number 7) and phosphorus (atomic number 15) belong to group 15 of the periodic table. Write their electronic configuration. Which of these will be more electronegative and why?
SOLUTION:Ans.: The electronic configurations of the two elements are :
Nitrogen (Z = 7) 2, 5 ; Phosphorus (Z = 15) 2, 8, 5
Nitrogen is more electronegative than phosphorus. The electronegativity decreases down the group with increase in size.
How does the electronic configuration of an atom relate to its position in the modern periodic table?
SOLUTION:Ans.: The electronic configuration of an atom helps us to predict the position of the atom in the periodic table.
(i) The number of the outermost shell corresponds to the period. For example, if the atom has second outermost shell, it belongs to second period and so on.
(ii) The number of electrons in the outermost shell corresponds to group in the periodic table.
In the modern periodic table, calcium (atomic number 20) is surrounded by elements with atomic number 12, 19, 21, and 38. Which of these have physical and chemical properties resembling calcium?
SOLUTION:Ans.: Only those elements are placed in the same group in which the gaps of atomic numbers are : 8, 8, 18, 18, 32. If we look at the atomic numbers of the elements that are listed, it becomes clear that the elements with atomic numbers 12, 20 (Ca), 38 fit into this pattern. They are placed in the same group and have also similar physical and chemical properties.
Q 23.Compare and contrast the arrangement of elements in Mendeleev's periodic table and the modern periodic table.
SOLUTION:Ans.: (i) In Mendeleev's periodic table the elements are arranged in the increasing order of atomic masses. On the other hand, in the modern periodic table the elements are arranged in the increasing order of atomic numbers which is more fundamental property than atomic mass.
(ii) In the Mendeleev periodic table, the position of the elements was in accordance with atomic mass. In the modern periodic table the position of the elements is governed by electronic configurations, which determine their properties.
(iii) In Mendeleev periodic table, the position of isotopes was not justified. In Modern periodic table, the classification is based on the atomic number and not atomic mass and hence the position of isotopes is fully justified.
(iv) Mendeleev periodic table had some anomalies on the basis of atomic mass. In modern periodic law, there are no misfits in terms of atomic number. For example, in Mendeleev's periodic table, potassium was having position lower than argon. But in Modern periodic table, this has been solved. Argon precedes potassium because argon has atomic number 18 and potassium has 19.
(v) The Mendeleev periodic table does not explain satisfactorily as to why the properties of the elements are repeated at regular intervals of 2, 8, 18 and 32. The modern periodic table explains this logically.
(vi) There is no clear cut separation of different types of elements in the Mendeleev's periodic table. In modern periodic table there is clear cut separation of different types of elements such as representative elements, noble gases, transition elements.
(vii) Mendeleev periodic table is not easy to remember. Modern periodic table is easy to remember.